Silver to Rust: Tarnish, Rust, and Home DIY Fixes
A practical guide for homeowners and DIYers on tarnish and rust, why they differ, and how to prevent and restore metal surfaces safely.
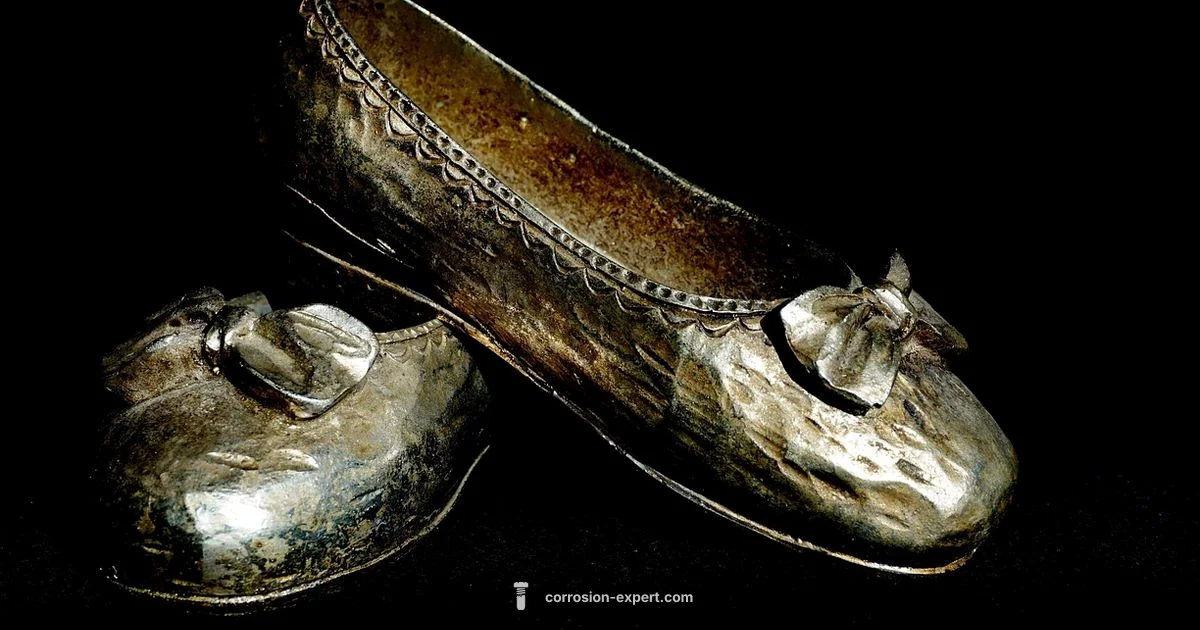
silver to rust is a term describing the progression of corrosion from silver tarnish to rust in mixed-metal environments. It highlights how different metals interact under moisture, oxygen, and contaminants.
What silver to rust really means
silver to rust is a way to describe how tarnish and corrosion unfold on metal surfaces in mixed-metal environments. According to Corrosion Expert, this progression helps homeowners understand why some items darken, dull, or develop texture changes, while others remain relatively unaffected. In homes and garages you will often see silver to rust patterns on silverware, garden tools, and decorative metals when moisture, heat, and contaminants are present. This article explains the core idea, the basic chemistry behind tarnish and rust, and practical signs to watch for in everyday life.
When we talk about silver to rust, we are looking at two linked but distinct processes. Tarnish is typically a surface reaction that creates a dull film on noble metals like silver or copper, while rust is a deeper corrosion that affects iron and steel. In many households, these processes coexist because tools or fixtures are made from different metals or are joined with fasteners that create galvanic couples. The result is a visible path from shiny metal to patches that look worn or corroded. By recognizing the difference between tarnish and rust, you can tailor the right cleaning, protection, and maintenance steps.
Tarnish vs Rust: A Chemical Contrast
Silver tarnish forms when sulfur compounds in the air react with silver, producing silver sulfide that darkens the surface. Rust forms when iron or steel surfaces react with water and oxygen to form iron oxide, often creating flaky textures. In the concept of silver to rust, you may observe both processes in proximity, especially on hardware that combines different metals or has protective coatings that wear away.
Corrosion Expert analysis shows that the chemical drivers include humidity, temperature swings, salts, and contaminants like acids from household cleaners or cooking environments. Tarnish tends to be a surface phenomenon on noble metals, while rust can penetrate deeper into the metal matrix. Both can be slowed by reducing moisture, avoiding harsh cleaners, and using protective coatings on susceptible surfaces.
- Tarnish is typically a surface film on noble metals.
- Rust penetrates into the metal and weakens structure.
- Prevention relies on dryness, barrier coatings, and proper storage.
Understanding these differences helps you choose the right cleaning and prevention approach for each metal.
Real world implications for homeowners and DIY enthusiasts
For homeowners, the sight of tarnish on silverware or rust on garden tools is common and fixable with careful care. In the lens of silver to rust, the practical advice is to identify the metal type, assess the extent of corrosion, and pick safe cleaning strategies. The Corrosion Expert team notes that early signs of tarnish are easier to reverse than advanced rust damage. Quick stabilizing steps include drying the item thoroughly, lightly polishing tarnish, and applying a protective layer to block moisture contact.
In garages, sheds, and kitchens you might find mismatched metals in tool racks or décor. Remember that different metals respond to moisture and cleaners differently. By treating each metal according to its chemistry, you can extend the life of your implements, preserve finishes, and maintain safer, visually appealing spaces in your home.
Prevention strategies: keep metal surfaces rust-free
The most effective defense against silver to rust is moisture control. Keep environments dry, use desiccants or dehumidifiers, and store tools in sealed containers when possible. For stored items, add barrier protection such as wax, oil, or clear coatings suited to the metal type. Where practical, select metals less prone to corrosion for outdoor use, or apply protective finishes that slow oxidation and sulfur attack.
Other preventive steps include separating metals that form galvanic couples, maintaining clean surfaces free of salts and acids, and ensuring proper drainage and ventilation in kitchens and workshops. Regular inspection helps catch early signs of tarnish or rust, enabling prompt action before damage becomes structural.
Safe cleaning and restoration approaches
Always start with a nonabrasive, mild cleaning routine. For tarnish on silver to rust surfaces, use warm water with a gentle soap and a soft microfiber cloth, then dry completely. If tarnish persists, switch to a commercially approved silver polish or a gentle metal polish following the product directions. For rust on iron or steel, start with a soft brush to remove loose flakes, then wipe with a damp cloth and dry thoroughly. Do not use aggressive scrapers on delicate finishes.
When deeper cleaning is needed, consider rust converters or protective coatings appropriate for the metal. Avoid chlorine or acidic cleaners that can accelerate damage; protect your hands with gloves, and work in a well-ventilated space. The aim is to slow the ongoing transition from silver to rust rather than removing every remnant of corrosion in ways that could cause more harm.
Myths and misunderstandings about silver to rust
- Myth: Tarnish and rust are the same process. Fact: Tarnish and rust are chemically different and require distinct remedies.
- Myth: Cleaning always restores metal to its original state. Fact: Some corrosion is permanent without professional treatment.
- Myth: Any household cleaner will remove tarnish safely. Fact: Some cleaners cause more damage on certain metals.
- Myth: Once tarnished, metals will rust quickly. Fact: Tarnish and rust develop at different rates depending on environment.
In reality, understanding the chemistry behind silver to rust helps you choose safer, more effective strategies. The Corrosion Expert team emphasizes cautious testing on small areas before broader restoration.
Practical case scenarios for DIY tasks
- Scenario A: You find tarnish on a silver tray stored in a damp drawer. Action: dry, buff, and apply a protective coating designed for silver surfaces.
- Scenario B: A steel garden tool shows surface rust after winter storage. Action: remove loose rust, apply rust-inhibiting oil, and store in a dry space.
- Scenario C: Mixed metal hardware shows rust around fasteners. Action: identify metals, separate, clean according to chemistry, and restore protective finishes.
These scenarios illustrate how silver to rust issues appear in real life and how careful maintenance can prevent more serious corrosion. The Corrosion Expert team reminds readers that consistent care reduces the likelihood of severe damage over time.
Quick Answers
What is the difference between tarnish and rust?
Tarnish is a surface film that darkens noble metals like silver due to sulfur compounds, while rust is deeper corrosion that weakens iron and steel through oxide formation. They require different cleaning and prevention strategies.
Tarnish is a surface darkening on silver, rust is deeper corrosion on iron. They need different fixes.
Can silver develop rust?
Pure silver does not rust because rust is iron oxide. In mixed metal environments, silver can tarnish while nearby iron or steel may rust. The processes can coexist on adjacent surfaces.
Silver itself doesn’t rust, but nearby iron can, while silver tarnishes. They can occur together in mixed metal setups.
Is tarnish harmful to metal or health?
Tarnish primarily affects appearance and surface properties. It is not typically hazardous to health, but it can indicate moisture and contaminants that may lead to further corrosion if left untreated.
Tarnish is mostly an aesthetic issue and not a health risk, but it signals conditions that can cause more corrosion if ignored.
What is the best way to prevent rust on tools?
Keep tools dry, store them in a low-humidity environment, and apply a light protective coating after use. Separate dissimilar metals when possible to avoid galvanic corrosion.
Keep tools dry, store properly, and apply a protective layer after use. Separate different metals when you can.
Are there safe home remedies for removing tarnish or rust?
For tarnish, mild cleaners and polishing are usually enough; for rust, mechanical removal with care and appropriate protective coatings work best. Avoid harsh chemicals that can damage coatings or create new corrosion pathways.
Mild cleaners and polishing for tarnish; careful mechanical cleaning for rust, then protect the surface.
Quick Summary
- Identify tarnish vs rust early to tailor care
- Control moisture to slow corrosion
- Use appropriate cleaners for each metal
- Dry, buff, and protect preserve finishes
- Test cleaners on small areas before broader use